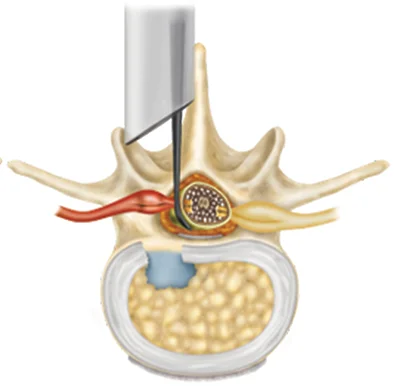
Most Common Treatment for Herniated Disc: Discectomy
If non-surgical therapies have failed, a discectomy surgery is typically recommended. A lumbar discectomy is a surgical procedure that is performed to relieve back and leg pain caused by a herniated disc. The goal of the procedure is to remove the portion of the disc that is pressing against the spinal nerves. However, after the portion of the disc is removed, there can be a hole (defect) left.